When we talk about specific gravity, we are exploring a simple but powerful idea in science. It helps us understand why some objects float while others sink, why oil stays on top of water, and how scientists compare different materials. In this article, you will learn everything about specific gravity in a clear, easy, and interesting way.
What Is Specific Gravity?
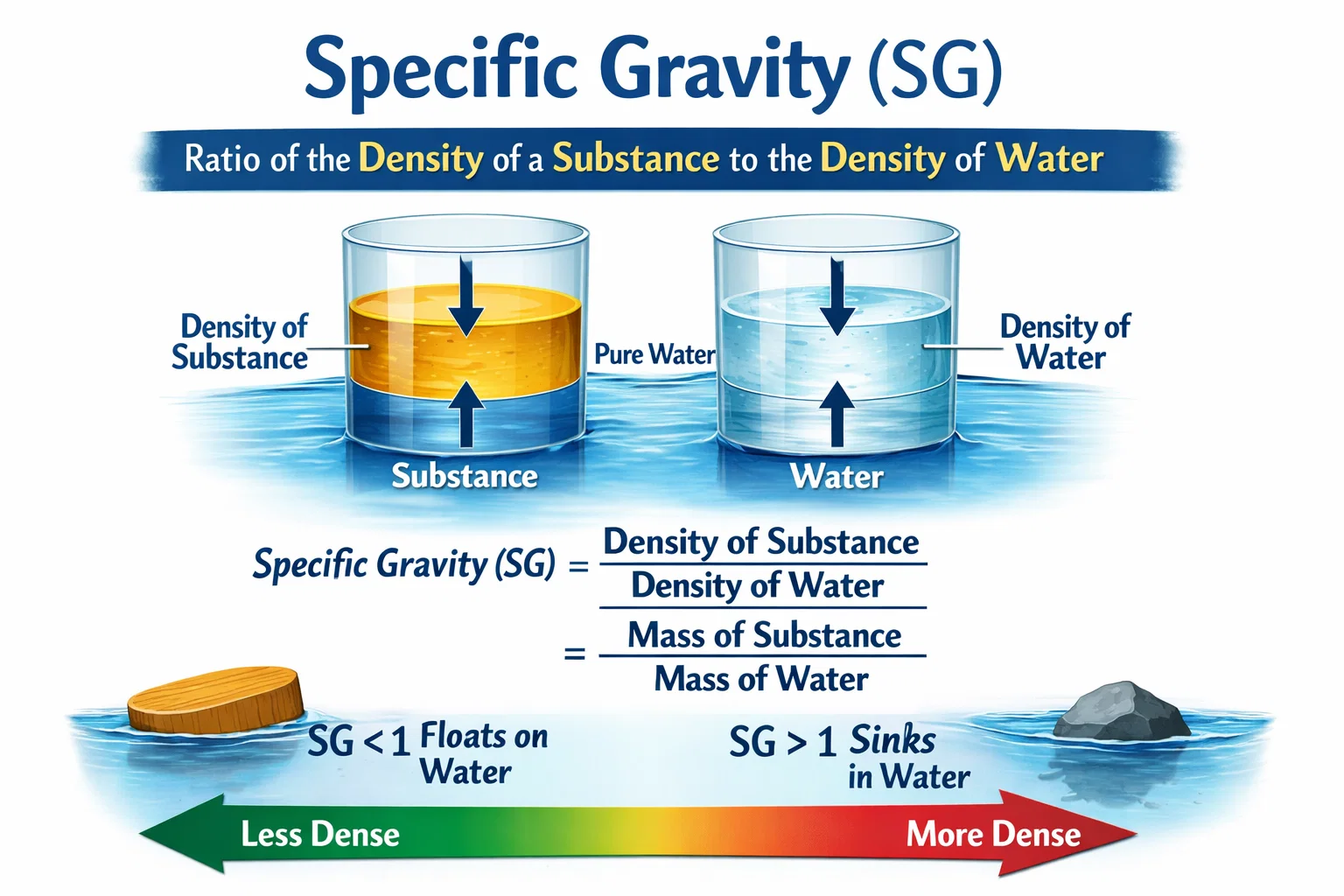
Specific gravity is a measure that compares the density of a substance to the density of water. In simple words, it tells us how heavy something is compared to water.
- If specific gravity = 1, the substance has the same density as water
- If specific gravity > 1, the substance is heavier than water
- If specific gravity < 1, the substance is lighter than water
For example:
- Wood has a specific gravity less than 1, so it floats
- Iron has a specific gravity greater than 1, so it sinks
So, you see, specific gravity helps us predict behavior in liquids very easily.
Why Specific Gravity Is Important
You might wonder, “Why do we even need to learn about specific gravity?” Well, it’s more useful than you think!
Here are some reasons:
- It helps in engineering and construction
- It is used in medicine and health tests
- It plays a role in chemistry experiments
- It helps in quality control of materials
- It explains natural phenomena like floating and sinking
In everyday life, specific gravity is quietly working behind the scenes.
The Formula of Specific Gravity
The formula of specific gravity is very simple:
Specific Gravity = Density of Substance ÷ Density of Water
Since water is the standard, its density is taken as 1 (in simple units). So, the formula becomes easy to use.
Example:
If a material has a density of 2, then:
- Specific gravity = 2 ÷ 1 = 2
That means the material is twice as dense as water.
Specific Gravity vs Density
Many students confuse specific gravity with density. Let’s clear that up!
| Feature | Specific Gravity | Density |
|---|---|---|
| Definition | Comparison with water | Mass per unit volume |
| Unit | No unit (dimensionless) | Has units (g/cm³, kg/m³) |
| Reference | Always compared to water | No reference needed |
| Use | Simple comparison | Detailed measurement |
So, while they are related, specific gravity is much easier to understand and use in quick comparisons.
How to Measure Specific Gravity
There are several ways to measure specific gravity, depending on the situation.
Common Methods:
- Hydrometer
- A floating device used in liquids
- Often used in labs and industries
- Balance Method
- Compare weight in air and water
- Pycnometer
- A special container used in laboratories
Each method helps find specific gravity accurately.
Examples of Specific Gravity in Daily Life
Let’s look at some everyday examples of specific gravity:
Floating Objects
- Ice floats on water because its specific gravity is less than 1
- Ships float even though they are heavy, because of their design
Kitchen Examples
- Oil floats on water (lower specific gravity)
- Honey sinks in water (higher specific gravity)
Nature Examples
- Dead Sea water allows people to float easily
- Rocks sink in rivers due to high specific gravity
Isn’t it amazing how specific gravity explains so many things around us?
Specific Gravity of Common Substances
Here is a helpful table showing specific gravity values:
| Substance | Specific Gravity |
|---|---|
| Water | 1.0 |
| Ice | 0.92 |
| Wood | 0.6 – 0.9 |
| Oil | 0.8 |
| Milk | 1.03 |
| Iron | 7.8 |
| Gold | 19.3 |
This table shows how different materials compare using specific gravity.
Applications of Specific Gravity in Science
Specific gravity is widely used in many fields of science.
In Chemistry
- Identifying unknown substances
- Checking purity of liquids
In Physics
- Studying buoyancy and fluid behavior
In Geology
- Identifying minerals and rocks
In Medicine
- Testing urine concentration
Clearly, specific gravity plays a key role in scientific discoveries.
Factors That Affect Specific Gravity
Even though specific gravity is a simple concept, some factors can affect it:
Temperature
- As temperature increases, density changes
- This can change specific gravity slightly
Pressure
- High pressure can increase density
Composition
- Mixtures behave differently than pure substances
So, it’s important to consider these factors when measuring specific gravity.
Fun Experiments to Understand Specific Gravity
Want to try something fun at home? Let’s explore specific gravity with easy experiments!
Egg Floating Experiment
What you need:
- Water
- Salt
- Egg
Steps:
- Put egg in plain water → it sinks
- Add salt and stir
- The egg starts to float
Why? Because adding salt increases the water’s specific gravity.
Common Mistakes About Specific Gravity
Here are some common misunderstandings:
- Thinking it has units (it does NOT)
- Confusing it with weight
- Ignoring temperature effects
- Assuming all heavy objects sink
Avoid these mistakes to better understand specific gravity.
Benefits of Learning Specific Gravity
Learning about specific gravity has many benefits:
- Improves science knowledge
- Helps in real-life problem solving
- Builds strong basics for higher studies
- Makes learning fun and practical
It’s a small concept with big importance!
Conclusion: Understanding Specific Gravity Made Easy
To sum it up, specific gravity is a simple yet powerful concept that helps us compare substances with water. It explains why objects float or sink, supports scientific research, and is used in many real-life applications.
From classrooms to laboratories, from kitchens to oceans, specific gravity is everywhere. Once you understand it, you’ll start noticing its role in daily life and that’s when science becomes truly exciting!
So next time you see something floating or sinking, just remember it’s all about specific gravity!